Research Overview
Dr. Carolyn Fredericks’ research uses advanced imaging tools to better understand how Alzheimer’s disease progresses through functional networks in the brain. She has a special interest in preclinical Alzheimer’s disease, sex differences in Alzheimer’s disease, and on less common Alzheimer’s variants. She and her research team seek to enable earlier detection of Alzheimer’s disease and identify new targets for therapy.
Dr. Fredericks’ research laboratory is located at Yale University in New Haven, Connecticut, USA. Her team is part of Yale’s Clinical Neuroscience Imaging Center (CNIC), a multidisciplinary group applying innovative imaging methods to the study of brain disease. Clinically, Dr. Fredericks sees patients with a variety of cognitive and behavioral concerns. She specializes in the diagnosis and treatment of neurodegenerative disorders.
Current Lab Projects
Sex Differences in Alzheimer’s Disease
Music in Alzheimer’s Disease
Parkinson’s, Dementia with Lewy bodies, and Psychosis
Click here for more information about these studies and how to get involved!
Giulia Lorenzon
Giulia is a postdoctoral researcher investigating how sex and reproductive factors influence brain network connectivity and tau pathology in Alzheimer’s disease (AD). Building on her PhD at Karolinska Institutet, where she examined AD and aging heterogeneity through MRI biomarkers, she now integrates multimodal neuroimaging (tau PET and functional MRI), plasma biomarkers, and deep clinical phenotyping in longitudinal cohorts of amnestic and non-amnestic AD to study how functional network organization predicts tau accumulation in men and women. She is also interested in how stress-related factors shape brain–body interactions. Ultimately, her work aims to uncover sex-specific vulnerabilities in AD and their underlying neural network mechanisms.
Wen-Xiang Tsai
Wen-Xiang is a doctoral researcher investigating how functional brain network organization changes from healthy aging to MCI and Alzheimer’s disease. He uses intrinsic timescale methods (including EMD) to examine how integration, segregation, and cross-system communication vary across frequency-specific networks. His goal is to identify timescale-selective network markers that support earlier detection and better characterization of Alzheimer’s disease.
Maya Agdali
Maya is a third year doctoral student within the lab studying how hormones influence the brain’s structural, functional, and vascular systems to shape sex-specific resilience or vulnerability to Alzheimer’s disease. Her work integrates multimodal neuroimaging approaches to characterize how testosterone and other sex hormones affect white matter microstructure, network connectivity, and cerebral blood flow across aging populations. By linking these to cognitive outcomes, she ultimately seeks to identify sex-specific neural pathways that may inform targeted strategies for early detection and intervention in AD.
Jordan Galbraith
Jordan is a third year MD/PhD student who is interested in understanding why women are at increased risk for Alzheimer's disease compared to men. Specifically, she leverages resting state functional MRI to study functional connectivity changes in mid-life and how they relate to changing hormones and cognition throughout the menopause transition. She also works with PET data to study sex differences in synaptic density loss in people with early-stage Alzheimer's disease.
Spencer Price
Spencer is interested in memory and cognitive control circuitry supporting language. They are investigating how differences in features of spoken language map onto changes in the brain's structural and functional connectivity in populations with primary progressive aphasia (PPA). Spencer hopes to better characterize network changes in PPA variants and improve our understanding of how the brain supports language functions.
Raina Vin
Raina Vin is a PhD student co-mentored by Dr. Carolyn Fredericks and Dr. Al Powers. She studies visual hallucinations in Parkinson’s disease and dementia with Lewy bodies, and uses magnetic resonance imaging, positron emission tomography, and graph theory to relate brain networks to behavior. She also leads the lab’s dementia with Lewy bodies study that enrolls individuals with dementia with Lewy bodies, Parkinson’s disease, and REM behavior disorder.
Alumni:
Hamid Abuwarda
Hamid Abuwarda was an MD/PhD student in the lab and graduated with his PhD in September of 2025! Hamid investigated how functional connectivity patterns can predict tau pathology distribution in preclinical Alzheimer's disease (AD). He found predictive features that could define novel, data-driven patient subgroups across both preclinical and atypical AD presentations. By characterizing how tau spreads in relation to functional connectivity within these newly identified subgroups, Hamid gained deeper insights into the differences in underlying disease trajectories.
What is Atypical Alzheimer’s Disease?
Most of us think of Alzheimer’s disease (AD) as primarily a disease of memory. Many of us have a friend or loved one who has personally experienced this most common “amnestic” form of Alzheimer’s disease, struggling more and more with short-term memory, having trouble finding their way on familiar roads, and asking family members repeated questions. And indeed, most of the time Alzheimer’s disease targets brain structures that specialize in supporting these functions. However, for reasons we don’t yet understand, Alzheimer’s sometimes targets brain structures and networks that are specialized for visuospatial processing (in which case we call it posterior cortical atrophy (PCA)), or for certain aspects of language (logopenic aphasia, also known as logopenic variant primary progressive aphasia (lvPPA)), or judgment and thinking (frontal variant Alzheimer’s disease (fvAD)).
Patients who suffer from one of these variants often search for what seem evasive answers. Diagnosis is often delayed. The initial symptoms may not immediately seem neurologic, or symptom progression may confuse original conclusions. Since experiences for these patients and their families are so different from what we usually understand as Alzheimer’s disease or dementia, it is often a very isolating experience. And while support groups exist, these are more often set up for people with amnestic Alzheimer’s disease and may be focused on different issues than those experienced by people with atypical Alzheimer’s disease. For instance, someone with logopenic aphasia who was once a social butterfly may now have trouble following conversation in social environments, leading to frustration, social anxiety, and self-isolation. Caregivers may also find it difficult to find other caregivers who truly understand. Hope may arise for some people with the prospect of engaging in a clinical trial—but inclusion criteria often exclude people with atypical presentations of Alzheimer’s. All this, along with a vision of advancing our knowledge of brain targets of these diseases to lead to development of new treatments, is why we study it.
Diagnosis is based on history, neurological examination, neuropsychological testing, neuroimaging scans such as magnetic resonance imaging (MRI) and positron emission tomography (PET) scans, spinal taps measuring cerebrospinal fluid (CSF) markers, and other tools available to clinicians. Criteria for meeting diagnosis are often discussed and published by consensus panels. Usually many measures go into arriving at a diagnosis, including some key clinical features:
Key clinical features of posterior cortical atrophy include difficulties with visual and visuospatial processing. Often the first symptoms are of visual discomfort—e.g., experiencing too much glare in the window when driving on a bright day, or too much light when confronted with headlights or other lights on the road at night. Patients also may struggle to find one item when too many other items surround it—e.g., when searching for ketchup on a crowded refrigerator shelf. They may have trouble putting on clothing—e.g., a sweater when a sleeve is inside out—or trouble making out checks because they are unable to make sense of each line. Many patients often struggle with basic aspects of visual processing such as form, motion, and color. As with other AD variants, patients with PCA often struggle with mood symptoms such as anxiety and have a degree of insight into their illness that they experience as “painful” (Benson et al. 1988). We believe these symptoms are an important part of the illness. In our lab we use Crutch et. al 2017 criteria to define PCA.
On top is an image a person with PCA has been asked to copy, which has been drawn below with difficulty.
Z-scores across cognitive domains, and raw Mini-Mental State Examination (MMSE) scores, are significantly worse in the posterior cortical atrophy (PCA) group than the healthy control (HC) group, but visuospatial function is disproportionately affected. Error bars represent standard deviation. (*p < 0.005). (Fredericks et al., 2019)
Key clinical features of logopenic aphasia include, first and foremost, symptoms in the realm of language. More specifically, patients with this variant struggle with particular aspects of language related to word-finding, repetition, and complex syntax. It’s hard for them to find words—and they often pause in the midst of a phrase as they struggle with this, or ultimately produce a word in which some sounds or syllables are inverted or simply incorrect. Because the part of the brain we use to rehearse complex sentences in our minds is affected early in lvPPA, they may struggle with repetition of complex phrases. They also struggle with sentences with complex syntax. Other forms of primary progressive aphasia exist, including nonfluent/agrammatic aphasia and semantic dementia. Symptoms sometimes overlap, making it difficult to determine which variant a person has. Logopenic aphasia typically shows similar pathology to amnestic AD (abnormal amyloid-β and tau) and is thus categorized as atypical Alzheimer’s; in contrast, the other variants of primary progressive aphasia do not have these pronounced pathological similarities and are not categorized as Alzheimer’s variants. In our lab we use Gorno-Tempini et. al 2011 criteria to define lvPPA.
People with variants of Alzheimer’s disease also have distinct patterns of pathology in the brain, as shown in the figure from Ossenkoppele et al. 2016. However, brain networks affected in these individuals are not well understood.
Why Study Atypical Alzheimer’s Disease?
We believe that we cannot understand Alzheimer’s disease without studying its less common variants. Our goals are to understand which brain networks are affected in these variants—including those brain networks responsible for common mood symptoms such as anxiety and irritability—and learn how these networks are impacted by tau pathology and how this progresses over time. We hope to use these measures as biomarkers for trials of future promising treatments, and that understanding more about critical brain structures affected early in these variants could lead to new ideas for treatments.
Research Projects
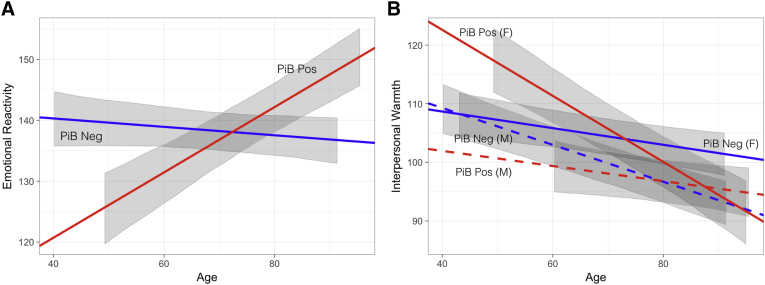
Our research has focused on brain networks—a collection of brain regions that seem to work in tandem to support a certain function—that are disrupted or enhanced in both preclinical Alzheimer’s disease and posterior cortical atrophy, as well as how these relate to cognitive and neuropsychiatric symptoms. We have shown that increased emotional reactivity is an early, significant feature in preclinical Alzheimer’s disease (see figure below), and that this may be related to higher connectivity in the insula and superior temporal sulcus, key brain regions involved in emotional and social processing (Fredericks et al. 2018).
Visualization of fixed effects modeling emotional reactivity and interpersonal warmth over time by amyloid status. Emotional reactivity increases longitudinally in cognitively normal PiB-positive individuals (β = 0.55, t = 4.0, P < .001) (A), whereas interpersonal warmth decreases over time in all participants, significantly more so in PiB-positive women (β = 0.61, t = 3.3, P = .001) (B). In (A), slopes are specified by the estimate of the fixed effect associated with age (for PiB-negative participants), and this fixed effect plus the fixed effect for the interaction of age by PiB positivity (for PiB-positive participants). Intercepts are specified by β0 for the model (for PiB-negative participants), and β0 plus the fixed effect of PiB positivity (for PiB-positive participants). (B) incorporates the fixed effect for sex and its interactions with PiB status and age to specify slopes and intercepts for PiB-negative and PiB-positive men and women. Gray ribbons represent the 95% CI of the fixed effects in the model, not incorporating residual variance or variance due to random effects, and are shown for the range of age values observed for each group. Abbreviation: PiB, [11C]-Pittsburgh compound B. (Fredericks et al., 2018, Figure 2)
Furthermore, people with posterior cortical atrophy have more depressive symptoms and personal distress than healthy individuals (see figure below), but preserved and even heightened empathy for others. We posit that the increased connectivity between brain regions in the salience network in PCA, demonstrated in Fredericks et al., 2019, relates to these symptoms and may relate to the disruption of the network that supports visuospatial functions. We also believe that the pulvinar nucleus, part of the thalamus (a central brain structure with far-ranging connections to other parts of the brain), may be involved. More research is warranted to understand why and whether these regions could be used in potential treatment targets.
Individuals with posterior cortical atrophy (PCA) show preserved empathetic concern and cognitive perspective-taking; they have increased levels of personal distress and score higher than healthy controls on depression screen (GDS). Error bars represent standard deviation. (*p ≤ 0.001). (Fredericks et al., 2019, Figure 3)